How to process data from clinical trials and their open label extensions PhUSE, Berlin, October 2010 Thomas Grupe and Stephanie Bartsch, Clinical Data. - ppt download

Phase II single arm open label multicentre clinical trial to evaluate the efficacy and side effects of a combination of gefitinib and methotrexate to treat tubal ectopic pregnancies (GEM II): study protocol

Effects of antiplatelet therapy on stroke risk by brain imaging features of intracerebral haemorrhage and cerebral small vessel diseases: subgroup analyses of the RESTART randomised, open-label trial - The Lancet Neurology
![PDF] Bias was reduced in an open-label trial through the removal of subjective elements from the outcome definition. | Semantic Scholar PDF] Bias was reduced in an open-label trial through the removal of subjective elements from the outcome definition. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/814ad6ccbfa9defca4d2b00c4672f9070cf6b8da/16-Figure1-1.png)
PDF] Bias was reduced in an open-label trial through the removal of subjective elements from the outcome definition. | Semantic Scholar
PLOS ONE: A Phase I, Open-Label Trial, Evaluating the Safety and Immunogenicity of Candidate Tuberculosis Vaccines AERAS-402 and MVA85A, Administered by Prime-Boost Regime in BCG-Vaccinated Healthy Adults

Study design schematic for the open-label extension study. AE, adverse... | Download Scientific Diagram
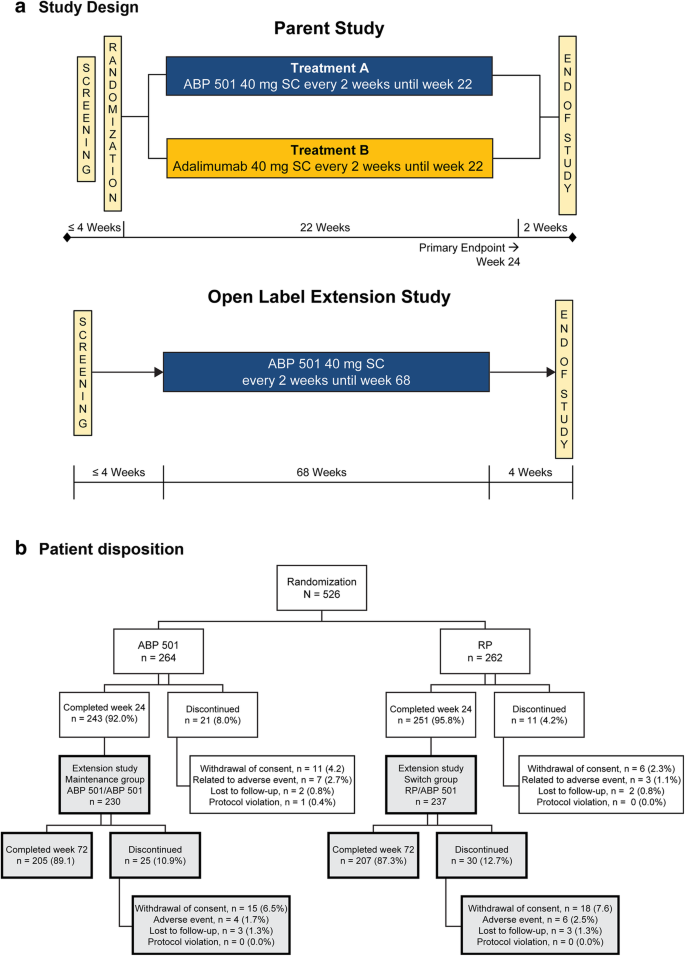
An open-label extension study to demonstrate long-term safety and efficacy of ABP 501 in patients with rheumatoid arthritis | Arthritis Research & Therapy | Full Text
PLOS Neglected Tropical Diseases: Efficacy and safety of available treatments for visceral leishmaniasis in Brazil: A multicenter, randomized, open label trial
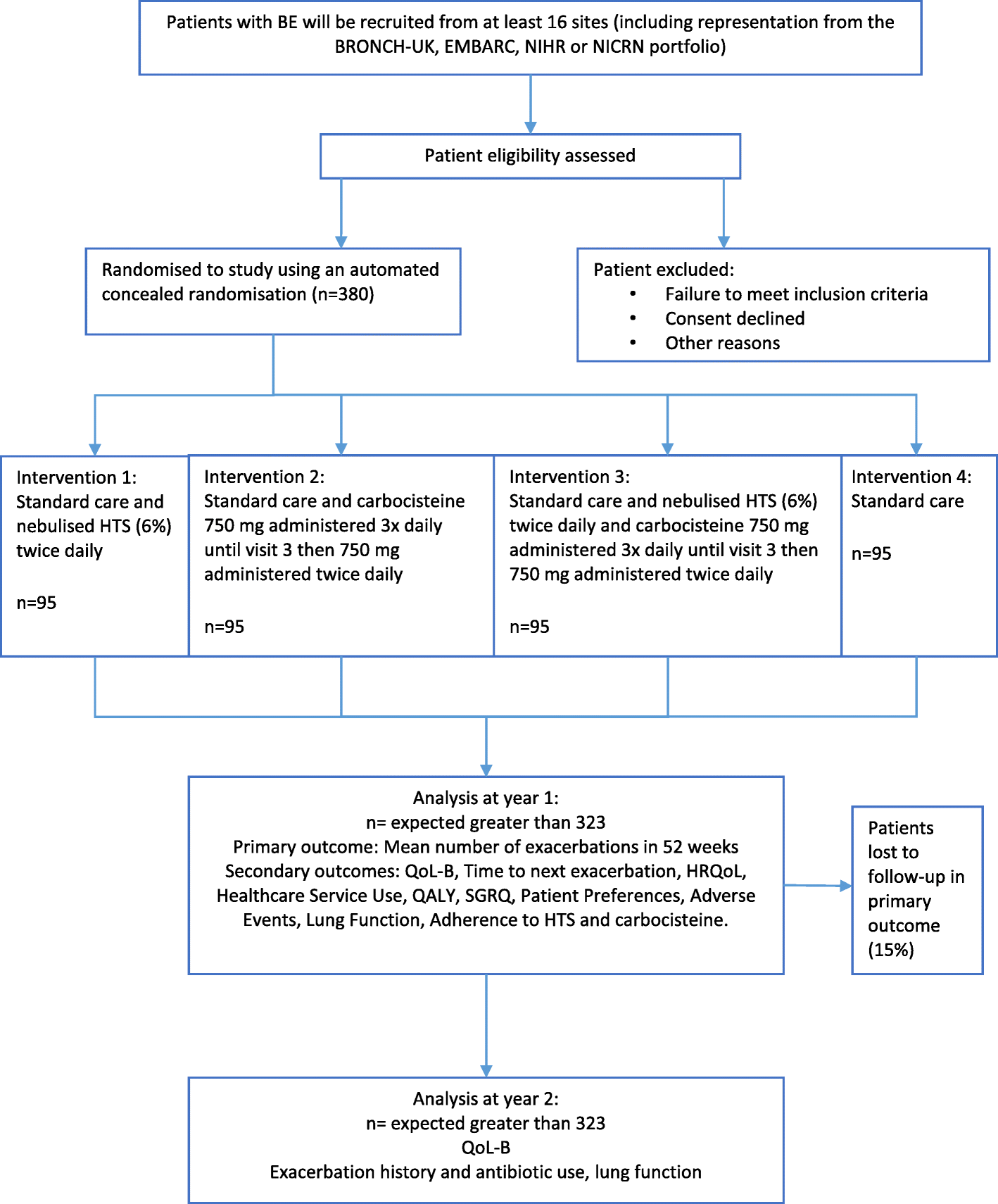
A 2 × 2 factorial, randomised, open-label trial to determine the clinical and cost-effectiveness of hypertonic saline (HTS 6%) and carbocisteine for airway clearance versus usual care over 52 weeks in adults

Rationale and design of a multi-center, open-label, randomised clinical trial comparing HIV incidence and contraceptive benefits in women using three commonly-used contraceptive methods (the ECHO study). | Semantic Scholar
![PDF] Open-label versus double-blind placebo treatment in irritable bowel syndrome: study protocol for a randomized controlled trial | Semantic Scholar PDF] Open-label versus double-blind placebo treatment in irritable bowel syndrome: study protocol for a randomized controlled trial | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/60afa7902fddea770036d6744ee84027545bd238/3-Figure1-1.png)
PDF] Open-label versus double-blind placebo treatment in irritable bowel syndrome: study protocol for a randomized controlled trial | Semantic Scholar

Single center, open label dose escalating trial evaluating once weekly oral ixazomib in ART-suppressed, HIV positive adults and effects on HIV reservoir size in vivo - eClinicalMedicine

An open-label randomized controlled trial of low-dose corticosteroid plus enteric-coated mycophenolate sodium versus standard corticosteroid treatment for minimal change nephrotic syndrome in adults (MSN Study) - Kidney International